
Division of Evolution, Infection & Genomics
School of Biological Sciences
Faculty of Biology Medicine & Health
The University of Manchester
danna.gifford@manchester.ac.uk
+44 (0)161 306 8074
Antibiotic resistance and evolutionary genomics
My research combines experimental, mathematical, and bioinformatic approaches to predict how and when bacteria will evolve antibiotic resistance under biologically-realistic growth conditions. The aim of this research is to develop a rational framework for designing combination therapies to suppress resistance evolution and regain usefulness of antibiotics where resistance has become wide-spread. This work is crucial for establishing combination therapies as a viable solution to the antibiotic resistance crisis. The project is funded through a UKRI Rutherford Fellowship, involving collaborations with Tobias Galla, Chris Knight, and Simon Lovell.
I am also involved in a multi-disciplinary BBSRC project to study the adaptive landscapes of antibiotic resistance evolution. This project combines wet-lab experimental evolution with mathematical models and advanced computer simulations to predict how population size and mutational target size interact to shape resistance evolution. I am also collaborating with Rok Krašovec on the effects of mutation rate plasticity on the evolutionary genomics of E. coli.
Other projects I’ve been involved with on include genomics and bioinformatics of antibiotic resistance evolution, e.g. species-level differences in beta-lactam resistance evolvability (with Victoria Furio), chromosomal versus plasmid-borne beta-lactam resistance evolvability (with Alvaro San Millan), mutational robustness (with Karl Heilbron) and the repeatability of resistance evolution (with Tom Vogwill).

In my DPhil, I studied evolutionary reversions of resistance and adaptive evolution in rifampicin resistant Pseudomonas aeruginosa. I demonstrated that the ability for a resistance reversion mutation to spread depends not only on the fitness of the resistant strain, but also its evolvability when faced with a non-preferred carbon source (L-serine). Different resistance mutations seem to confer a different ability to adapt to this substrate, putatively due to interactions between rpoB and a transcriptional regulator involved in L-glycine and L-serine metabolism (PA2449, now known as gcsR). Highly evolvable competitors can also spur revertants to adapt more.
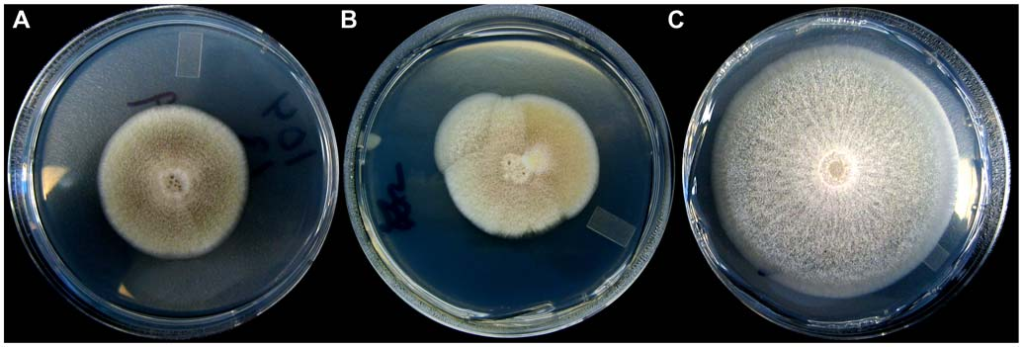
In my MSc work, I studied compensatory adaptation of Aspergillus nidulans in a complex medium. We found that the number of major fitness jumps during adaptive walks is independent of starting fitness—populations undergo just two major changes in fitness, on average.